![]()
Convolutional neural network feature learning
The power of CNN & DL in arrhythmia prediction
![]()
Unerring classing/ unerring classification
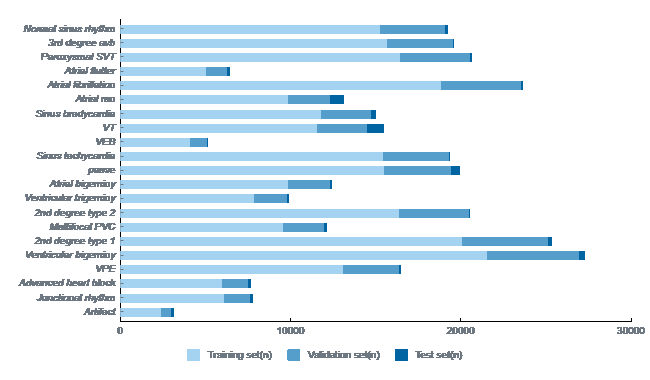
Detection & Validation of 21 categorizations of arrhythmias
![]()
Higher exactitude / higher precision
Accuracy score of 98% and a Precision score of 95% & growing
![]()
Certified ratification
Affirmed by MD American board-certified panel of Physicians
![]()
Substantial training dataset
Trained over a million data sets that are manually annotated by the American CCT/CRAT techs.
![]()
Hybrid prediction model
Built in fusion with Computer vision and Artificial neural network to detect arrhythmias
![]()
Manifold device affinity
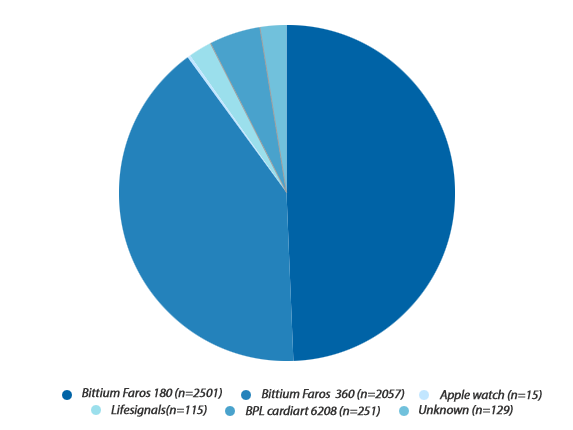
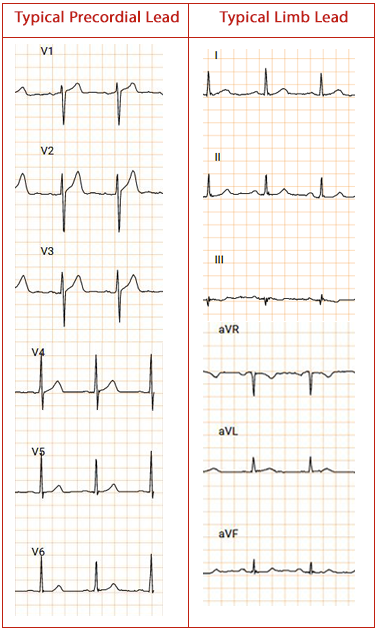
Works across a diversified set of digital ECG data sets; such as Holters, MCT, Events & 12 lead
![]()
HIPAA cloud infrastructure
Runs on secure HIPAA cloud infrastructure, enabling easy integration and customisation for IDTF’s & Wearables
Infuse RHYTHM AI application engine into your process to make more accurate detection, classification of 21 categorizations of cardiac arrhythmias and noise filters using machine learning algorithms. Amplify your team’s capabilities besides preventing costly medical events with a modular, user-friendly platform. We bring the powerful advantage of enterprise grade AI application for Holters, MCT, Events, 12 Lead ECG and Wearable biosensors to simplify and automate complex tasks.
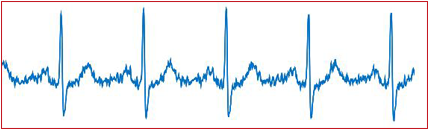
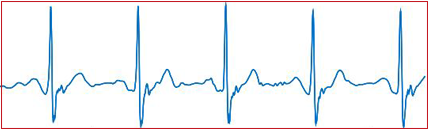
The conception of machine learning became engrossing in almost every sector as the healthcare industry stumbles its application as well. We are in devoir of an automated edifice that would administer the detection of assorted arrhythmias from the captured ECGs at the same time be mindful of the signal quality and analogous artifacts.
The approach we have adopted here is the implementation of Deep Learning for arrhythmia detection through a fresh approach utilizing discriminative visualization to enhance transparency and the interpretability of the Deep Neutral Network in an effort to imitate “Human eye interpretation”.
Data Collection
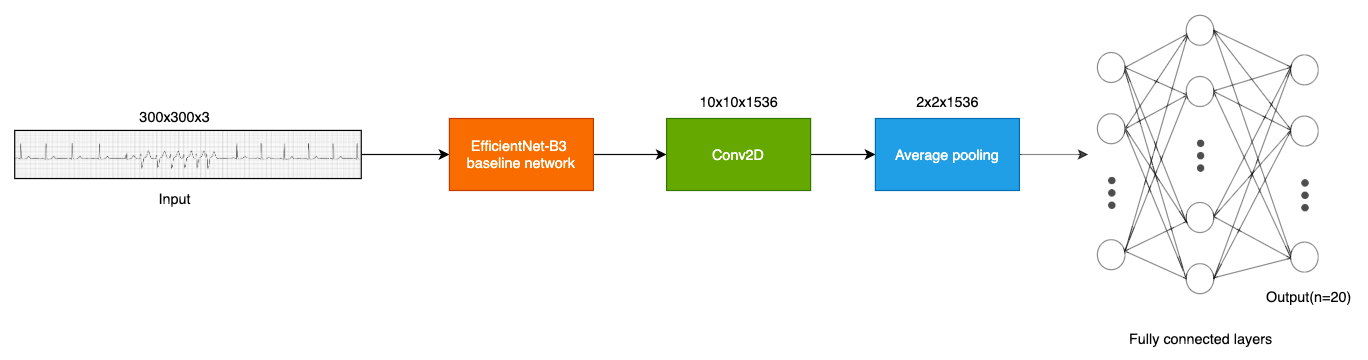
Data pipeline and model
1. Signal regularization: The ECG data from discrepant scales pertinent to their respective hardware are standardized to mV signal
2. Befitting lead selection:Felicitous data channel is designated just in case channel information is proven to be enigmatic or inaccessible
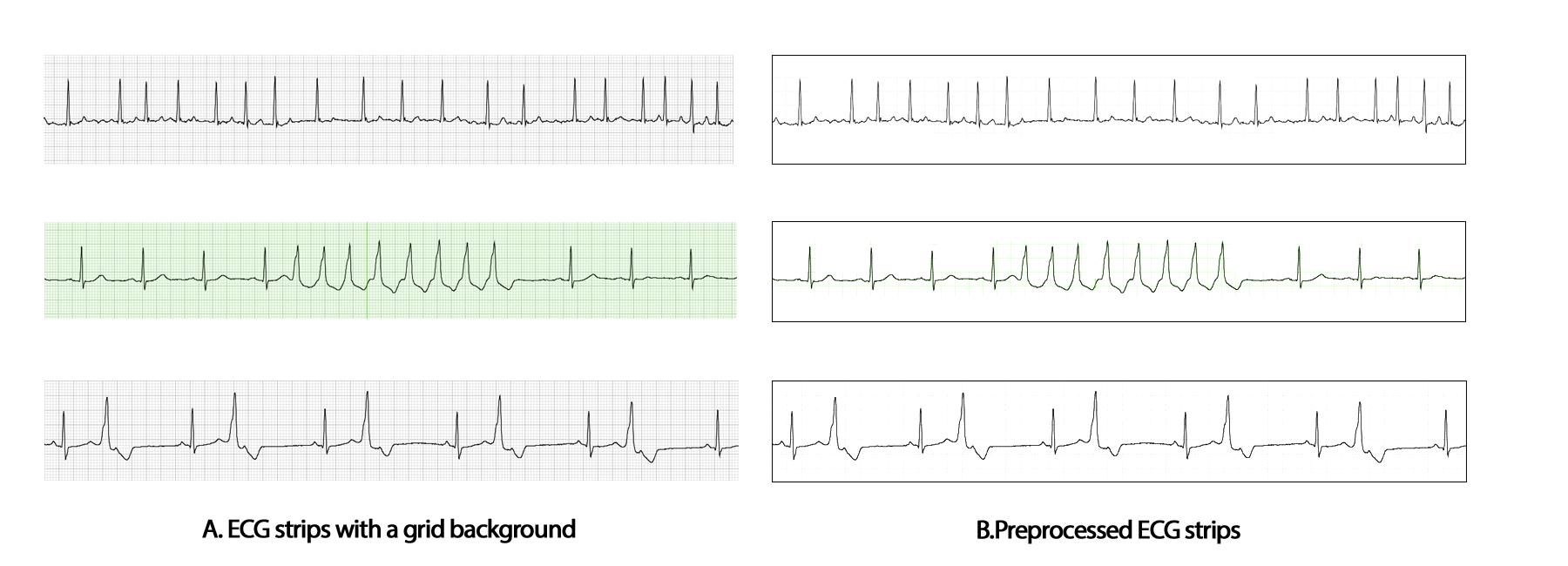
3. ECG strip production and dynamic cropping: Transformation of raw data for One-Dimensional to Two-Dimensional raw data transformation for classification.

Request For Services
Find out more about how we can help your organization navigate its next. Let us know your areas of interest so that we can serve you better.
Click here